Film coating
Mastering formulation and the coating process
The tablet coating process
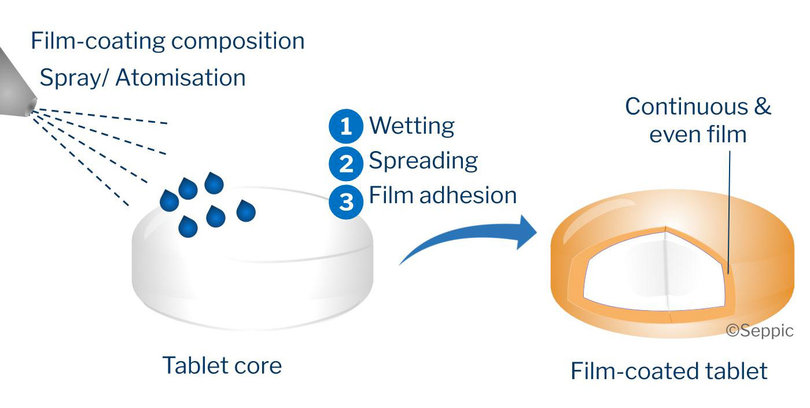
The coating process results in the formation of a thin layer of polymer around the tablet. This layer, 20 to 100 µm thick, represents approximately 1 to 10% of the tablet’s initial weight.
Dispersion
Coating technology involves having an expansive knowledge of ingredients and their interactions, expertise in formulation and in the various coating processes, in order to produce a smooth, regular and homogeneous film that completely covers the surface of the tablet. This requires the interaction between three elements:
- The tablet core, designed to withstand the mechanical stresses applied during the coating process and to facilitate the adhesion of the film to the tablet’s surface. The tablet’s shape strongly influences the mixing characteristics in the film-coating turbine and the uniformity of the final film on the tablets.
- Formulation of the coating, with its composition and viscosity.
- The coating process, and equipment, which strikes a delicate balance between atomization of the coating dispersion, and drying. The final quality of the film-coated tablets relies on critical process parameters.
Expertise in coating formulation
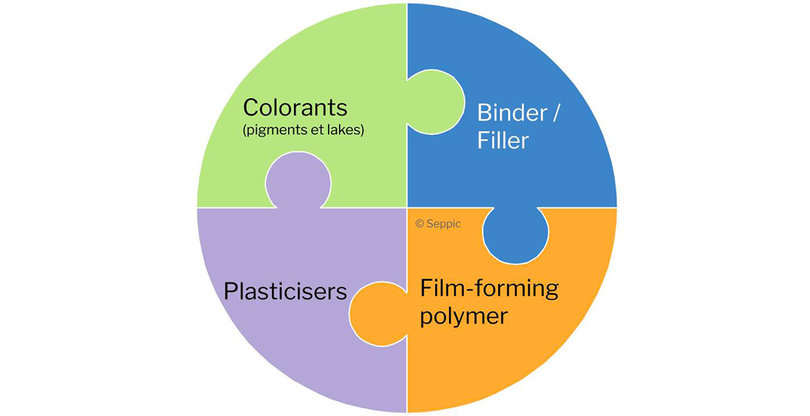
The film-coating formulation combines four key ingredients:
- A film-forming polymer, which provides the mechanical strength of the film. The most widely used polymers in non-functional film coating are cellulose derivatives such as hypromellose.
- Plasticizers, used to improve the flexibility of the film formed and prevent it from cracking or breaking. They work by weakening the attraction between the polymer molecules to make the film more malleable.
- Coloring agents, which increase opacity, protect from light, and color the film.
- Fillers, which can be added to reduce tack and improve slip, or to adjust the content of insoluble materials in the coating composition.
Seppic draws on in-depth knowledge of these different parameters of the coating stage and their interconnections, and its expertise in formulation, to develop robust, efficient, and ready-to-use formulations to facilitate the coating process by its customers. These formulations combine a film-forming polymer, plasticizer, and fillers, either with or without coloring agents, with appropriate grades selected and optimized proportions.